The study of carbon dioxide (CO2) is crucial in various scientific and industrial fields, from climate science to chemical engineering. One of the most fundamental tools for understanding the behavior of CO2 under different conditions is the phase diagram of CO2. This diagram provides a visual representation of the states of CO2—solid, liquid, and gas—under varying temperatures and pressures. By examining the phase diagram of CO2, scientists and engineers can predict how CO2 will behave in different environments, which is essential for applications ranging from carbon capture and storage to refrigeration systems.
Understanding the Phase Diagram of CO2
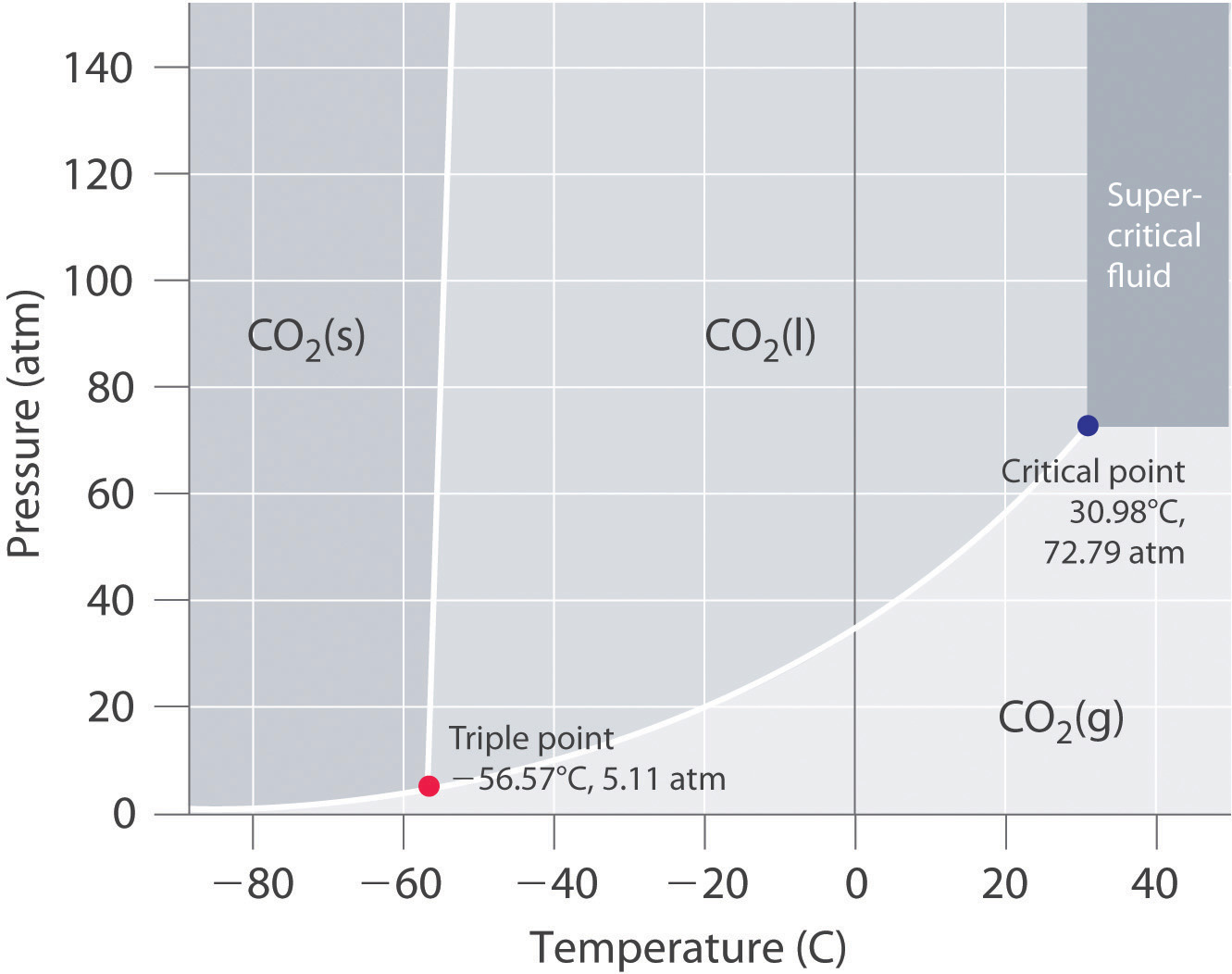
The phase diagram of CO2 is a graphical representation that shows the conditions under which CO2 exists in its different phases. The diagram typically plots pressure on the y-axis and temperature on the x-axis. Key features of the phase diagram include:
- The triple point, where the solid, liquid, and gas phases coexist.
- The critical point, where the liquid and gas phases become indistinguishable.
- The sublimation curve, which separates the solid and gas phases.
- The fusion curve, which separates the solid and liquid phases.
- The vaporization curve, which separates the liquid and gas phases.
The Triple Point of CO2
The triple point of CO2 is a critical point on the phase diagram where the solid, liquid, and gas phases coexist in equilibrium. For CO2, the triple point occurs at a temperature of approximately -56.6°C (or 216.55 K) and a pressure of about 5.11 bar. At this point, any small change in temperature or pressure can cause CO2 to transition between its solid, liquid, and gas states. Understanding the triple point is essential for processes that involve the phase transitions of CO2, such as in carbon capture and storage technologies.
The Critical Point of CO2
The critical point of CO2 is another crucial feature on the phase diagram. It marks the conditions above which the liquid and gas phases of CO2 become indistinguishable, forming a supercritical fluid. For CO2, the critical point occurs at a temperature of approximately 31.1°C (or 304.25 K) and a pressure of about 73.8 bar. Above the critical point, CO2 exhibits unique properties that make it useful in various applications, such as supercritical fluid extraction and enhanced oil recovery. The supercritical state allows CO2 to dissolve materials like a liquid while having the diffusivity of a gas, making it an effective solvent.
Applications of the Phase Diagram of CO2
The phase diagram of CO2 has numerous practical applications across various industries. Some of the key applications include:
- Carbon Capture and Storage (CCS): Understanding the phase behavior of CO2 is crucial for developing efficient CCS technologies. By knowing the conditions under which CO2 can be stored as a liquid or supercritical fluid, engineers can design systems that minimize leakage and maximize storage capacity.
- Refrigeration Systems: CO2 is used as a refrigerant in various cooling systems. The phase diagram helps in designing these systems by providing the necessary temperature and pressure conditions for CO2 to transition between its liquid and gas states efficiently.
- Supercritical Fluid Extraction: In this process, CO2 in its supercritical state is used to extract compounds from materials. The phase diagram guides the selection of operating conditions to ensure that CO2 remains in the supercritical state, enhancing the extraction efficiency.
- Enhanced Oil Recovery (EOR): CO2 is injected into oil reservoirs to increase oil recovery. The phase diagram helps in determining the optimal conditions for CO2 injection to ensure it remains in a supercritical state, which improves its ability to displace oil from the reservoir.
Experimental Techniques for Studying the Phase Diagram of CO2
Studying the phase diagram of CO2 involves various experimental techniques that allow scientists to observe and measure the phase transitions under different conditions. Some of the commonly used techniques include:
- Differential Scanning Calorimetry (DSC): This technique measures the heat flow associated with phase transitions. By monitoring the heat absorbed or released during the transition, scientists can determine the temperature and pressure conditions at which phase changes occur.
- X-ray Diffraction (XRD): XRD is used to study the crystal structure of solid CO2. By analyzing the diffraction patterns, researchers can identify the phases present and the conditions under which they form.
- Raman Spectroscopy: This technique provides information about the molecular vibrations of CO2 in different phases. By analyzing the Raman spectra, scientists can distinguish between the solid, liquid, and gas phases and study the phase transitions.
Challenges and Future Directions
While the phase diagram of CO2 provides valuable insights into its behavior, there are still challenges and areas for further research. Some of the key challenges include:
- Accuracy of Measurements: Precise measurement of temperature and pressure is crucial for accurate phase diagram construction. Advances in instrumentation and measurement techniques can improve the accuracy of phase diagrams.
- Complex Mixtures: In many industrial applications, CO2 is not present in pure form but as part of a mixture with other gases or liquids. Understanding the phase behavior of CO2 in these mixtures is more complex and requires further study.
- Dynamic Conditions: Many industrial processes involve dynamic conditions where temperature and pressure change rapidly. Studying the phase behavior of CO2 under these dynamic conditions is essential for optimizing process efficiency.
🔍 Note: Future research should focus on developing more accurate measurement techniques and understanding the phase behavior of CO2 in complex mixtures and dynamic conditions.
Conclusion
The phase diagram of CO2 is a powerful tool for understanding the behavior of CO2 under different conditions. By providing a visual representation of the phase transitions, the diagram helps scientists and engineers design efficient systems for carbon capture and storage, refrigeration, supercritical fluid extraction, and enhanced oil recovery. The triple point and critical point are particularly important features that guide the selection of operating conditions for these applications. While there are challenges in accurately measuring and understanding the phase behavior of CO2, ongoing research and advancements in experimental techniques will continue to enhance our knowledge and improve industrial processes.
Related Terms:
- co2 phase diagram engineering toolbox
- phase diagram of co2 system
- phase diagram of co2 pdf
- triple point of co2
- phase diagram of carbon dioxide
- phase diagram of carbon